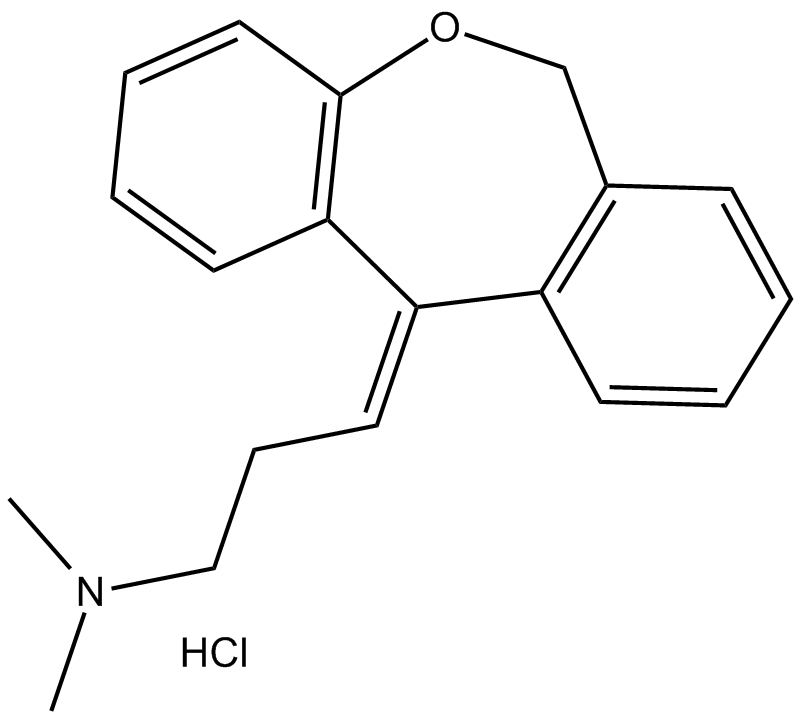
Doxepin (hydrochloride)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Doxepin, a tricyclic antidepressant, is a potent histamine H1 antagonist [1]. The histamine receptors belong to a family of G protein–coupled receptors activated by their primary endogenous ligand histamine. The H1 receptor has been expressed in smooth muscles, vascular endothelial cells, heart, and the central nervous system [2].
In vitro: Doxepin was a moderately potent competitive inhibitor of serotonin uptake in human blood platelets in vitro, with an inhibitory constant Ki of ~0.2 μM. Doxepin (100 μM) rapidly increased the efflux of serotonin from platelets [1].
In vivo: In rats and dog, oral administration of doxepin was well absorbed and quickly appeared in the blood. Numerous metabolites of doxepin were observed in liver and in urine, only doxepin and demethyl doxepin were found in the rat brain[3]..
Clinical trials: Doxepin has been marketed under many brand names worldwide. The oral topical formulations of doxepin are FDA-approved for the treatment of patients with major depressive disorder, primary insomnia, chronic urticaria, and anxiety [4,5,6].
References:
[1] Lingjrde O. Effect of doxepin on uptake and efflux of serotonin in human blood patelets in vitro[J]. Psychopharmacology, 1976, 47(2): 183-186.
[2] Hill S J, Ganellin C R, Timmerman H, et al. International Union of Pharmacology. XIII. Classification of histamine receptors[J]. Pharmacological reviews, 1997, 49(3): 253-278.
[3] Hobbs D C. Distribution and metabolism of doxepin[J]. Biochemical pharmacology, 1969, 18(8): 1941-1954.
[4] Hajak G, Rodenbeck A, Voderholzer U, et al. Doxepin in the treatment of primary insomnia: a placebo-controlled, double-blind, polysomnographic study[J]. Journal of Clinical Psychiatry, 2001, 62(6): 453-463.
[5] Feighner J P, Cohn J B. Double-blind comparative trials of fluoxetine and doxepin in geriatric patients with major depressive disorder[J]. The Journal of clinical psychiatry, 1985, 46(3 Pt 2): 20-25.
[6] Goldsobel A B, Rohr A S, Siegel S C, et al. Efficacy of doxepin in the treatment of chronic idiopathic urticaria[J]. Journal of allergy and clinical immunology, 1986, 78(5): 867-873.
| Storage | Store at -20°C |
| M.Wt | 315.8 |
| Cas No. | 1229-29-4 |
| Formula | C19H21NO·HCl |
| Synonyms | Sinequan |
| Solubility | ≥15.85 mg/mL in DMSO; ≥23.7 mg/mL in H2O; ≥26.5 mg/mL in EtOH |
| Chemical Name | 3-(dibenz[b,e]oxepin-11(6H)-ylidene)-N,N-dimethyl-1-propanamine, monohydrochloride |
| SDF | Download SDF |
| Canonical SMILES | CN(C)CC/C=C1C2=C(C=CC=C2)COC3=C/1C=CC=C3.Cl |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构