3MB-PP1

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
3MB-PP1 is a cell-permeable, potent, ATP-competitive, and highly selective inhibitor of polo-like kinase.
Human polo-like kinase 1 (PLK1) plays dominant role in mitosis and the maintenance of genomic stability. PLK1 is overexpressed in human tumours and exihibits prognostic potential in cancer, indicating its involvement in carcinogenesis and its potential as a therapeutic target [2].
In vitro: 3MB-PP1 treatment significantly changed the DNA state. In TbPLKas cells, after treatment with 3MB-PP1 for 9 h, cells at all cell cycle stages showed an increase in detached new flagella when compared with vehicle control-treated samples [1]. The IC50 value of 3MB-PP1 against Ptoas kinase activity was 120 nM. 3MB-PP1 was not able to significantly inhibit Pti1 and MPK2. 3MB-PP1 significantly potentiated the interactions of Ptoas with AvrPto and AvrPtoB1–387[3].
References:
[1] Lozano-Núez A, Ikeda K N, Sauer T, et al. An analogue-sensitive approach identifies basal body rotation and flagellum attachment zone elongation as key functions of PLK in Trypanosoma brucei[J]. Molecular biology of the cell, 2013, 24(9): 1321-1333.
[2] Strebhardt K, Ullrich A. Targeting polo-like kinase 1 for cancer therapy[J]. Nature reviews cancer, 2006, 6(4): 321-330.
[3] Salomon D, Bonshtien A, Mayrose M, et al. Bypassing kinase activity of the tomato Pto resistance protein with small molecule ligands[J]. Journal of Biological Chemistry, 2009, 284(22): 15289-15298.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 295.4 |
| Cas No. | 956025-83-5 |
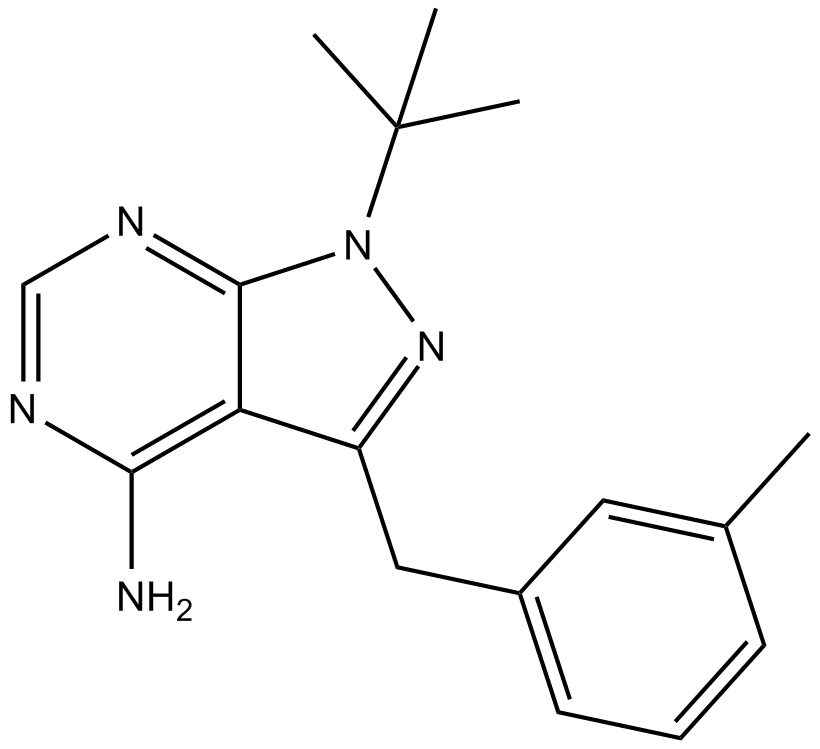
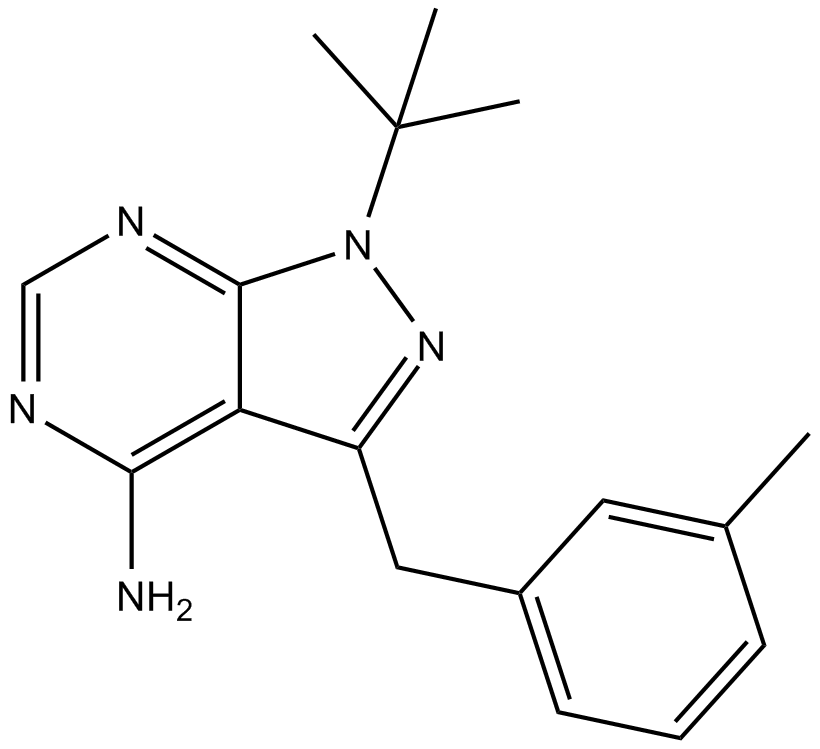
| Formula | C17H21N5 |
| Solubility | ≤2mg/ml in ethanol;20mg/ml in DMSO;25mg/ml in dimethyl formamide |
| Chemical Name | 1-(1,1-dimethylethyl)-3-[(3-methylphenyl)methyl]-1H-pyrazolo[3,4-d]pyrimidin-4-amine |
| SDF | Download SDF |
| Canonical SMILES | NC1=C(C(CC2=CC=CC(C)=C2)=NN3C(C)(C)C)C3=NC=N1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Cell experiment:[1] | |
|
Cell lines |
Trypanosoma brucei (T. brucei) cells expressing only analogue-sensitive TbPLK (TbPLKas) |
|
Reaction Conditions |
0.5, 1 or 5 μM 3MB-PP1 for 12 h incubation |
|
Applications |
The growth of the TbPLKas cells was strongly inhibited at 1 and 5 μM, with a clear growth defect appearing 6 h after the addition of 3MB-PP1. At this point the cells ceased to divide for the duration of the experiment. This result showed that TbPLKas cells treated with at least 1 μM 3MB-PP1 did not undergo cytokinesis within the first cell cycle. TbPLKas cells treated with 500 nM drug grew at ∼50% the rate of control cells. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Lozano-Núñez A, Ikeda KN, Sauer T, et al. An analogue-sensitive approach identifies basal body rotation and flagellum attachment zone elongation as key functions of PLK in Trypanosoma brucei. Molecular Biology of the Cell, 2013, 24(9): 1321-1333. |
|
化学结构