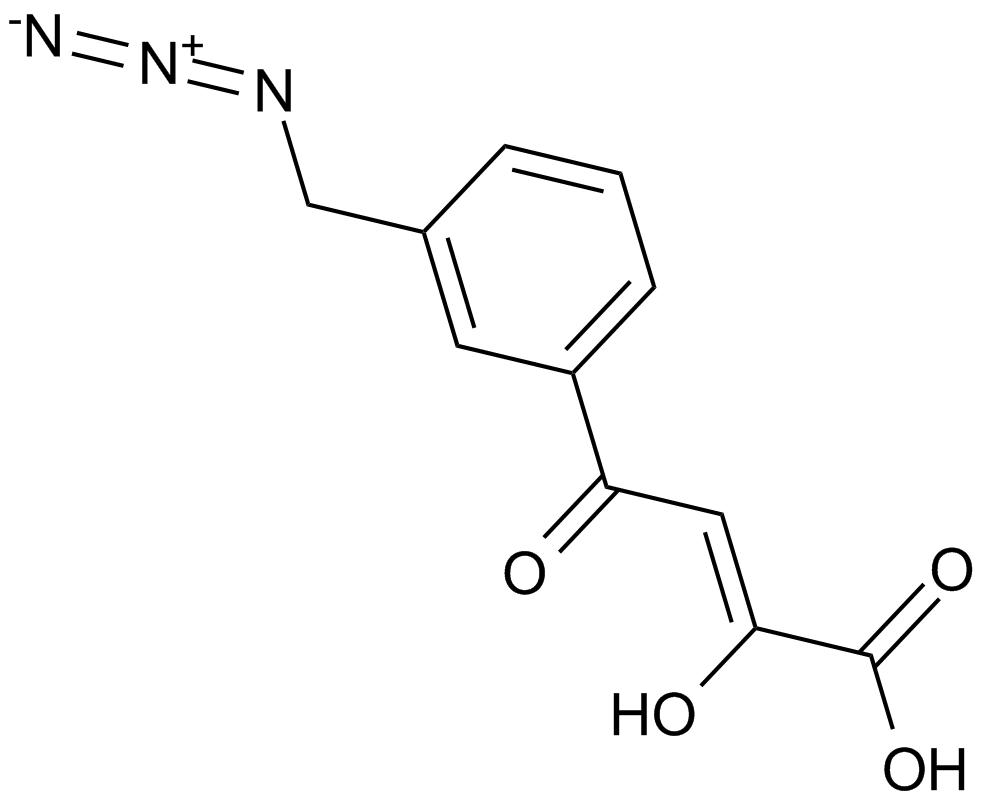
HIV-1 integrase inhibitor

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
HIV-1整合酶抑制剂用于抗HIV,其IC50值为0.33 μM[1],此抑制剂能靶向HIV-1整合酶,并抑制其活性,用于治疗HIV感染、AIDS以及其它类似的将逆转录病毒基因组整合到宿主染色体上的疾病。
HIV-1整合酶是源自Pol基因产物C末端部分的一段32 KDa的蛋白质,其由HIV产生以使HIV的基因能够整合到感染细胞的DNA中[2],其与噬菌体整合酶和逆转录病毒预整合复合体(PIC)中的一种关键组分也有所不同[3]。HIV-1整合酶由3个独立结构域组成,功能域—N末端域(NTD)、催化核心域(CCD)以及C末端域(CTD)。HIV-1整合酶通过多步反应行使功能,包括2个催化反应:前病毒DNA 3’末端内切核酸加工(termed 3’processing)和3’ 末端加工的病毒DNA整合到细胞DNA(称为链转移)中[4]。
人类免疫缺陷病毒(HIV)是获得性免疫缺陷综合症(AIDS)的病原体[5],因此HIV整合酶是抗HIV药物有希望的靶点。HIV-1整合酶抑制剂的药物设计包括整合酶链转移抑制剂(INSTIs)、抑制LEDGF/p75-整合酶相互作用和整合酶结合抑制剂,链转移抑制是最直观明显并易于推行至今的。Mg2+和Mn2+是整合期重要的辅助因子,因此去除这些辅助因子(如通过螯合作用)将导致整合酶的功能丧失[6]。竞争性抑制剂直接与病毒DNA竞争结合整合酶,从而抑制3’末端加工[7]。此过程中抑制剂通过靶向结合DNA从而完全阻断活性位点。INSTIs通过螯合二价金属离子(Mg2+),特异性紧密结合到IN的DNA末端,这是由催化三联体协调的,如位于CCD上的酶活化位点DDE基序[8]。
Merck Sharp & Dohme Limited开发了雷特格韦,成功完成了INSTI治疗的开发[9]。S/GSK1349572是由ViiV/Shinongi开发的整合酶抑制剂,已于2011年进入三期临床试验。此药物具有良好的耐受性,目前来看比雷特格韦和埃替格韦药效更好[10]。
参考文献:
1. Loizidou EZ et al. Analysis of binding parameters of HIV-1 integrase inhibitors: correlates of drug inhibition and resistance. Bioorg Med Chem. 2009, 17(13):4806-18.
2. Cocohoba, J; Dong, BJ. "Raltegravir: the first HIV integrase inhibitor". Clinical therapeutics.2008, 30(10): 1747–65.
3. Mouscadet, JF; Delelis, O; Marcelin, AG; Tchertanov, L. "Resistance to HIV-1 integrase inhibitors: A structural perspective". Drug resistance updates : reviews and commentaries in antimicrobial and anticancer chemotherapy.2010, 13(4-5):139–50.
4. Fan, X; Zhang, FH.et al."Design of HIV-1 integrase inhibitors targeting the catalytic domain as well as its interaction with LEDGF/p75: a scaffold hopping approach using salicylate and catechol groups". Bioorganic & Medicinal Chemistry.2011,19 (16): 4935–52.
5. Pommier, Yves.et al. "Integrase inhibitors to treat HIV/Aids". Nature Reviews Drug Discovery.2005, 4 (3): 236–248.
6. Pendri, A.et al. "New first and second generation inhibitors of human immunodeficiency virus-1 integrase". Expert opinion on therapeutic patents. 2011,21 (8): 1173–89.
7. Chen, X; Tsiang, M, Yu, F, Hung, M, Jones, GS, Zeynalzadegan, A, Qi, X, Jin, H, Kim, CU, Swaminathan, S, Chen, JM. "Modeling, analysis, and validation of a novel HIV integrase structure provide insights into the binding modes of potent integrase inhibitors". Journal of Molecular Biology. 2008, 380 (3): 504–19.
8. Mouscadet, JF. et al."Resistance to HIV-1 integrase inhibitors: A structural perspective". Drug resistance updates : reviews and commentaries in antimicrobial and anticancer chemotherapy.2010,13(4-5):139–50.
9. McColl, DJ; Chen, X. "Strand transfer inhibitors of HIV-1 integrase: bringing IN a new era of antiretroviral therapy". Antiviral Research,. 2010,85 (1): 101–18.
10. Barnhart, Matthew,James Shelton."A better state of ART improving antiretroviral regimens to increase global access to HIV treatment". Journal of AIDS and HIV Research. 2011, 3 (4): 71–78.
| Storage | Store at -20°C |
| M.Wt | 247.21 |
| Cas No. | 544467-07-4 |
| Formula | C11H9N3O4 |
| Solubility | Soluble in DMSO |
| Chemical Name | (Z)-4-[3-(azidomethyl)phenyl]-4-hydroxy-2-oxobut-3-enoic acid |
| SDF | Download SDF |
| Canonical SMILES | C1=CC(=CC(=C1)CN=[N+]=[N-])C(=CC(=O)C(=O)O)O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Description | HIV-1整合酶抑制剂是HIV-1整合酶的抑制剂,用于抗HIV。 | |||||
| 靶点 | HIV-1 integrase | |||||
| IC50 | ||||||
化学结构