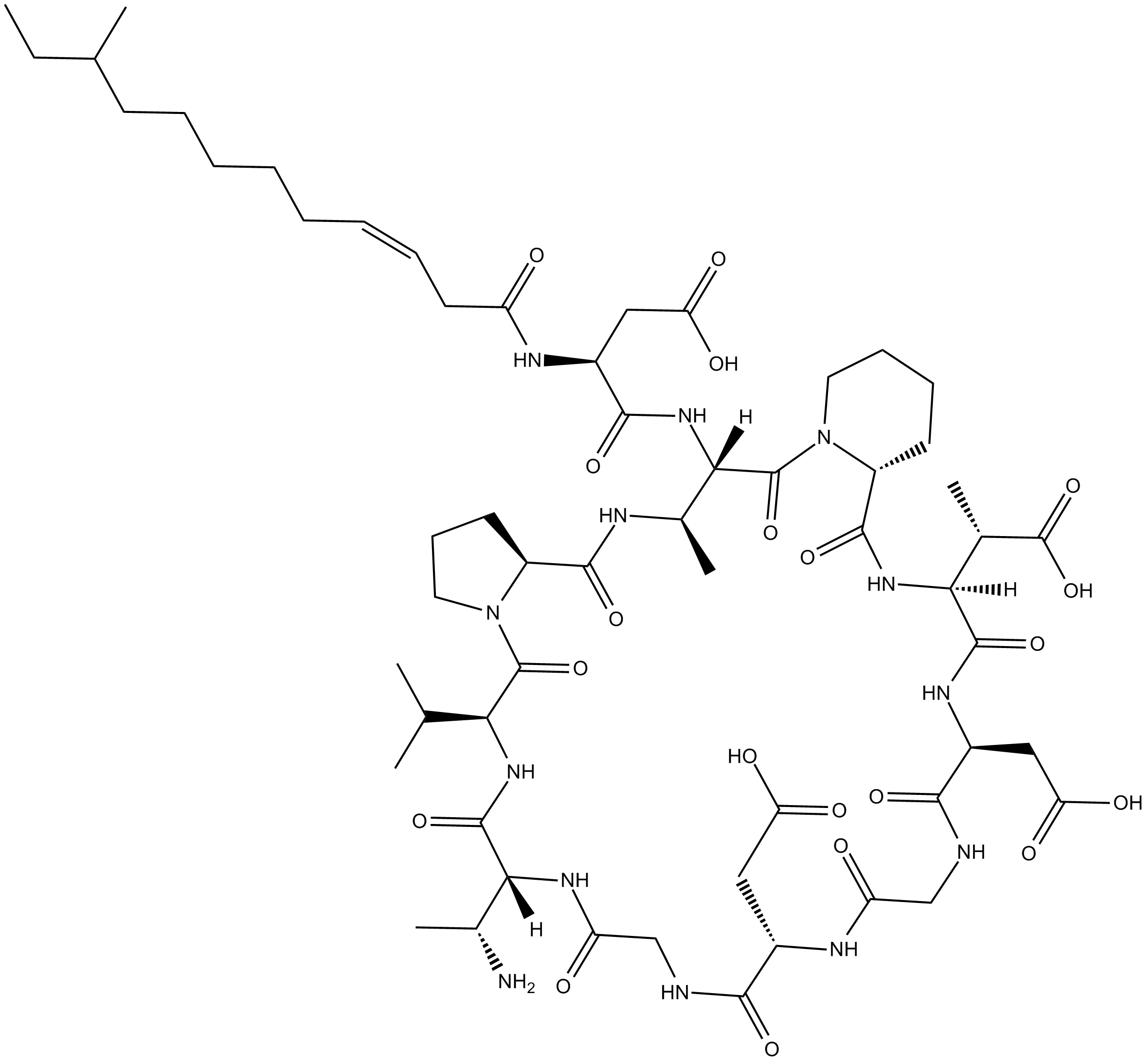
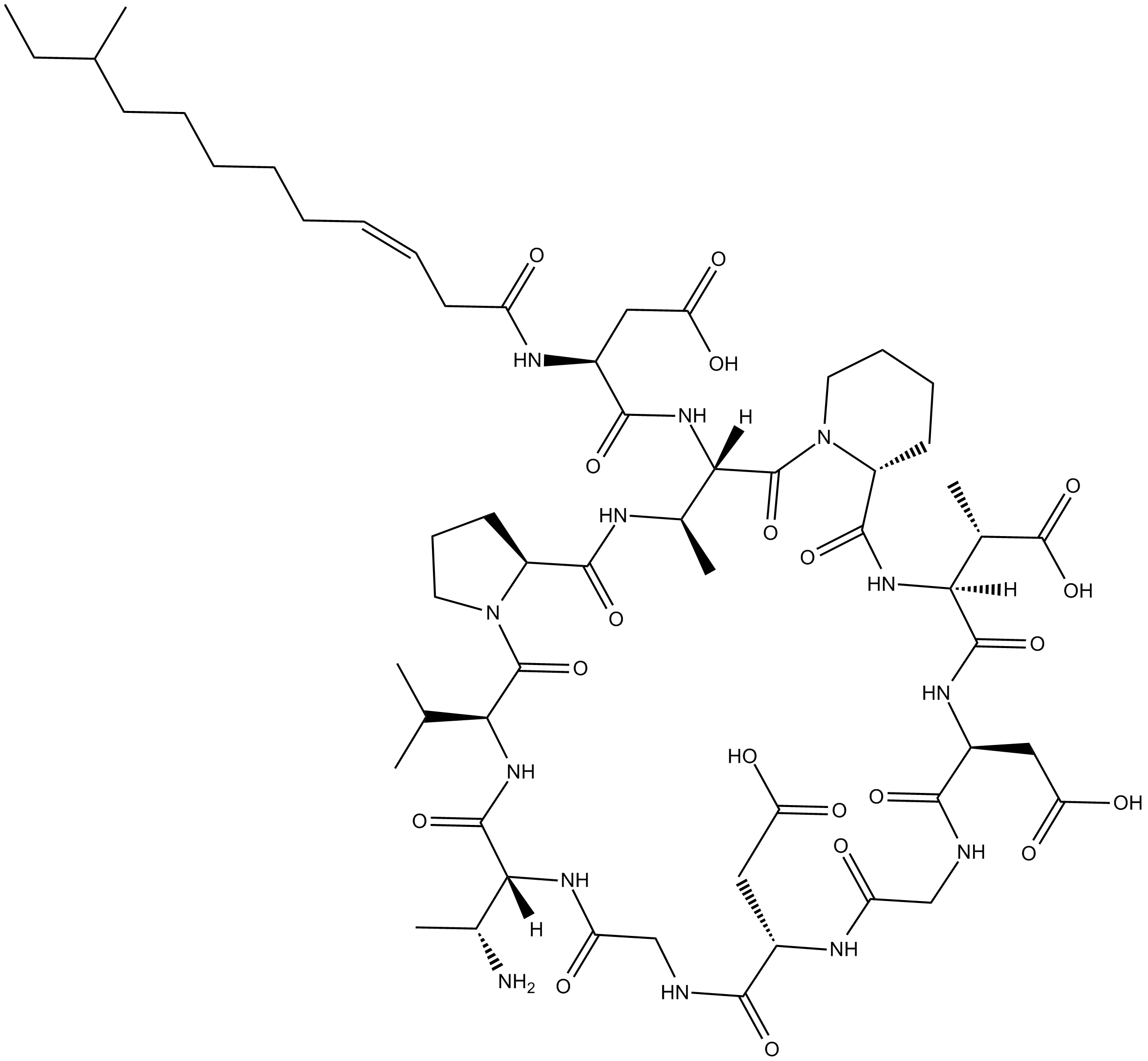
Amphomycin

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Amphomycin is a natural antibacterial lipopeptide.
Cyclic lipopeptides are a promising class of natural products with antibiotic properties. Cyclic lipopeptides are amphiphilic molecules, composed of a fatty acid tail linked to a short oligopeptide which form a macrocylic ring structure.
In vitro: In previous study, Calf brain endoplasmic reticulum membranes were incubated with varying concentrations of GDP-mannose in the presence and absence of amphomycin, results showed no significant difference in apparent Km for GDP-mannose. However, the Vmax was reduced in the presence of amphomycin as compared with in its absence. Moreover, when mannosylphosphoryldolichol synthase activity was measured in the presence of amphomycin, the shape of the substrate velocity curve changed from a rectangular hyperbola to a sigmoid [1].
In vivo: The PK of lipopeptides, the semi-synthetic amphomycin analogues, were evaluated in mice and rats following single i.v. and oral administration. Following oral administration at 50 mg/kg, plasma concentrations of amphomycin analogues were <0.3-0.9 μg/mL, indicating that oral availability was low. Following i.v. administration (5-10 mg/kg), the majority of lipopeptides demonstrated a long half-life, low clearance and a volume of distribution indicative of extracellular penetration. The long half-life and low clearance indicated that drug serum concentrations remained above the target minimal inhibitory concentration levels for significant periods of time. When combined with the potent efficacy against Gram-positive organisms, the results supported further development of these lipopeptide analogues towards clinical evaluation [2].
Clinical trial: Up to now, amphomycin is still in the preclinical development stage.
References:
[1] D. K. Banerjee. Amphomycin inhibits mannosylphosphoryldolichol synthesis by forming a complex with dolichylmonophosphate. The Journal of Biological Chemisty 264(4), 2024-2028 (1989).
[2] Pasetka CJ, Erfle DJ, Cameron DR, Clement JJ, Rubinchik E. Novel antimicrobial lipopeptides with long in vivo half-lives. Int J Antimicrob Agents. 2010 Feb;35(2):182-5.
| Physical Appearance | An off-white to fawn solid |
| Storage | Store at -20°C |
| M.Wt | 1290.4 |
| Cas No. | 1402-82-0 |
| Formula | C58H91N13O20 |
| Solubility | Soluble in DMSO |
| Chemical Name | 2,2'-((6S,9R,15S,21S,24S,26aR,33S,34R,36aS)-9-((R)-1-aminoethyl)-33-((2S)-3-carboxy-2-((Z)-10-methyldodec-3-enamido)propanamido)-24-((S)-1-carboxyethyl)-6-isopropyl-34-methyl-5,8,11,14,17,20,23,26,32,36-decaoxotetratriacontahydro-1H,5H-pyrido[1,2-a]pyrrol |
| SDF | Download SDF |
| Canonical SMILES | O=C([C@@H]1N(CCCC1)C([C@@](NC([C@@H](NC(C/C=C\CCCCCC(CC)C)=O)CC(O)=O)=O)([C@H](NC([C@@H]2CCCN2C([C@@H](NC([C@@]([H])([C@H](N)C)N3)=O)C(C)C)=O)=O)C)[H])=O)N[C@@]([C@@H](C(O)=O)C)(C(N[C@H](C(NCC(N[C@@H](CC(O)=O)C(NCC3=O)=O)=O)=O)CC(O)=O)=O)[H] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构