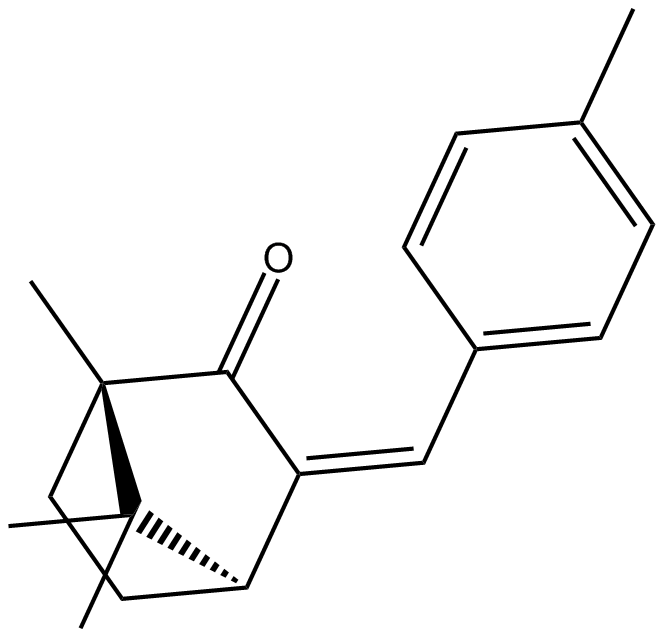
4-Methylbenzylidene camphor

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
4-Methylbenzylidene camphor (4-MBC) is an ultraviolet light blocker used in cosmetics and sunscreen preparations that also has estrogenic activities.
Since the estrogen receptor (ER) ligand type can influence transactivation, it is important to obtain information on molecular actions of nonclassical ER agonists.
In vitro: Previous results of competitive binding assays using cytosolic protein preparations from Xenopus hepatocytes demonstrated that 4-MBC could weakly bind to the ER. In addition, 4-MBC at a 100 micromol/L was not able to replace estradiol from the receptor completely [1].
In vivo: The estrogen target gene expression in uterus of Long Evans rats after exposure to 4-MBC was studied. Results showed that 4-MBC could altere steady-state levels of mRNAs encoding for ER, progesterone receptor (PR), and androgen recepto in uterus of 12-wk-old offspring. To evaluate sensitivity to estradiol (E2), offspring were injected with E2, and killed 6 h later. Acute up-regulation of PR and IGF-I and down-regulation of ER and androgen receptor by E2 were reduced in 4-MBC treated rats dose-dependently [2].
Clinical trial: In a 2-week single-blinded study, 15 young males and 17 postmenopausal females, were assigned to daily whole-body topical application of 2 mg per cm2 of 4-MBC basic cream formulation. Maximum plasma concentrations were 20 ng/mL for both females and men. 4-MBC was detectable in urine. The reproductive hormones FSH, LH were unchanged but minor differences in testosterone levels were observed between the 2 wk [3].
References:
[1] Klann A, Levy G, Lutz I, Müller C, Kloas W, Hildebrandt JP. Estrogen-like effects of ultraviolet screen 3-(4-methylbenzylidene)-camphor (Eusolex 6300) on cell proliferation and gene induction in mammalian and amphibian cells. Environ Res. 2005 Mar;97(3):274-81.
[2] S. Durrer, K. Maerkel, M. Schlumpf, et al. Estrogen target gene regulation and coactivator expression in rat uterus after developmental exposure to the ultraviolet filter 4-methylbenzylidene camphor. Endocrinology 146(5), 2130-2139 (2005).
[3] N. R. Janjua, B. Mogensen, A. M. Andersson, et al. Systemic absorption of the sunscreens benzophenone-3, octyl-methoxycinnamate, and 3-(4-methyl-benzylidene) camphor after whole-body topical application and reproductive hormone levels in humans. Journal of Investigative Dermatology 123, 57-61(2004).
| Storage | Store at -20°C |
| M.Wt | 254.4 |
| Cas No. | 36861-47-9 |
| Formula | C18H22O |
| Synonyms | Enzacamene|Eusolex 6300|4-MBC|Neo Heliopan MBC|Parsol 500|Uvinul MBC 95 |
| Solubility | insoluble in H2O; ≥10 mg/mL in DMSO; ≥111.2 mg/mL in EtOH |
| Chemical Name | 1,7,7-trimethyl-3-[(4-methylphenyl)methylene]-bicyclo[2.2.1]heptan-2-one |
| SDF | Download SDF |
| Canonical SMILES | CC1=CC=C(/C=C2[C@H]3CC[C@@](C)(C3(C)C)C/2=O)C=C1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构